Asia’s pharma and biotech sector is continuing to gain momentum, driven by global licensing deals, expanding clinical pipelines, platform technologies and growing investment in areas such as oncology, cardiometabolic diseases, diagnostics and drug delivery.
Drawing on the Financial Times’ 2026 ranking of the high-growth companies in Asia-Pacific, this list of the top 7 fastest-growing pharma and biotech companies in Asia for 2026 highlights companies that have shown strong growth while advancing programs across high-value therapeutic and technology areas. Take a look at company revenues, growth trajectory and recent activity, including regulatory milestones such as approvals and IND clearances, as well as notable dealmaking, licensing agreements, partnerships and investments.
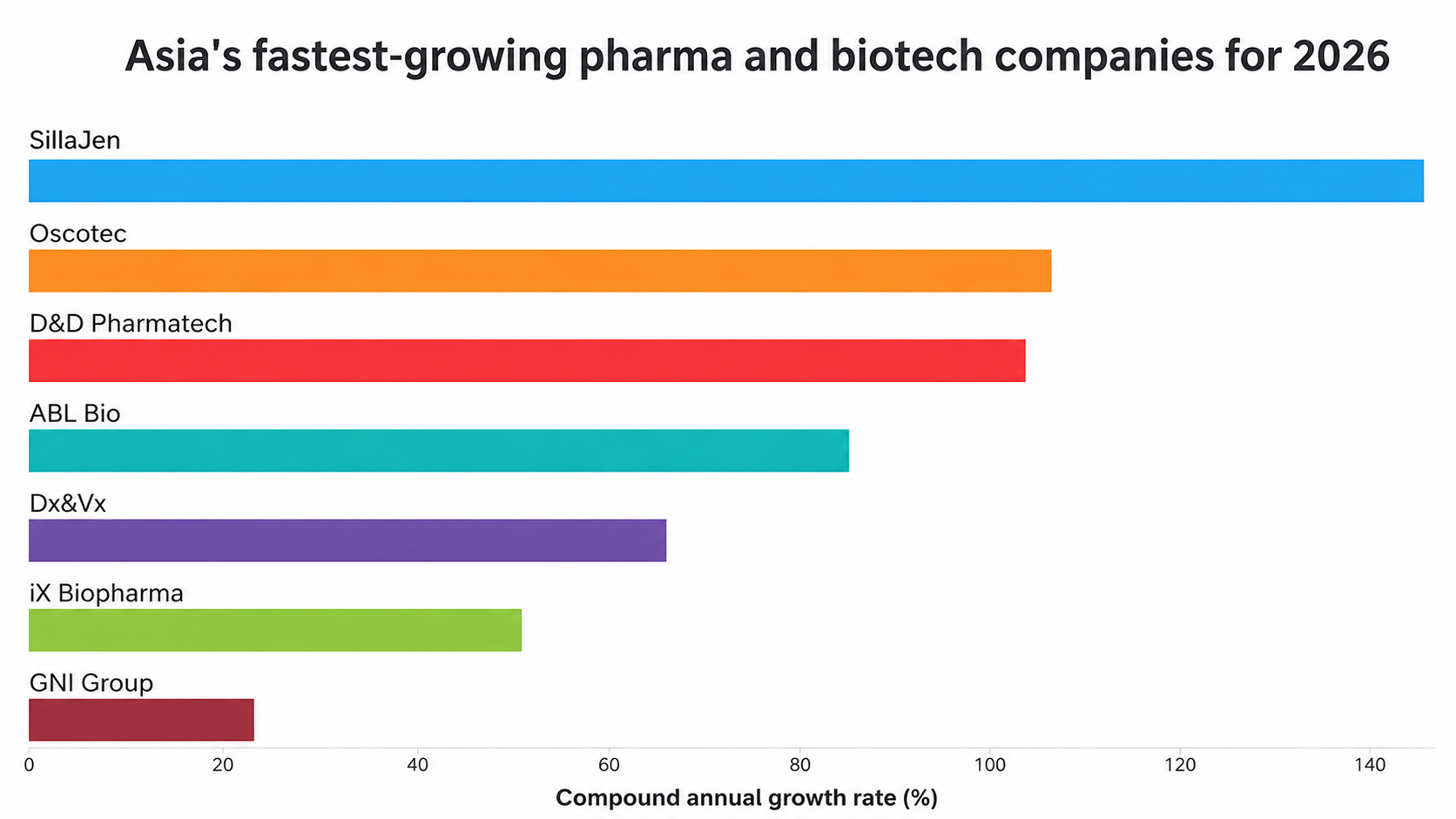
1. SillaJen
Location: South Korea
Overall rank: 25
Absolute growth rate (%): 1,443.72
Compound annual growth rate (%): 148.99
Revenue 2024: $2.88 million
Revenue 2021: $220,000
Number of employees: 45
Year founded: 2006
SillaJen is a South Korea-based biotechnology company focused on developing oncolytic virus-based immunotherapies for cancer. The company has 45 employees, and its 2024 revenue amounted to $2.88 million, growing at a compound growth rate of 148.99% since 2021. The company’s pipeline is centered on engineered viral immunotherapy candidates. Recently, SillaJen secured FDA approval for an IND amendment evaluating its anticancer drug BAL0891 in combination with the immune checkpoint inhibitor tislelizumab, supported by human vascularized organoid efficacy data generated through a collaboration with San Diego, US-based Qureator.
2. Oscotec
Location: South Korea
Overall rank: 50
Absolute growth rate (%): 771.38
Compound annual growth rate (%): 105.78
Revenue 2024: $24.94 million
Revenue 2021: $3.41 million
Number of employees: 51
Year founded: 1998
Oscotec is a South Korean biotech company focused on drug discovery and development in immunology and oncology. The company is closely associated with lazertinib, the EGFR-mutant NSCLC therapy commercialized through Yuhan/Janssen as part of the Rybrevant plus Lazcluze/Lecraza combination, which has secured approvals across major markets including the US, Europe, Japan and China. Oscotec had a strong 2025, reporting consolidated revenue of 99.84 billion won ($67.7 million) and operating income of 52.08 billion won ($35.3 million), helped by milestone payments and royalties tied to lazertinib’s global expansion. Oscotec’s revenue increased 193.5% from 34.01 billion won ($24.94 million) in 2024. Oscotec’s pipeline also includes ADEL-Y01, a tau-targeting antibody co-developed with ADEL that was licensed to Sanofi in December 2025 in a deal worth up to $1.04 billion. In March 2026, Oscotec entered an exclusive global option-to-license agreement with Yatiri Bio to develop and commercialize denfivontinib, also known as SKI-G-801, for acute myeloid leukemia (AML). The deal pairs Oscotec’s multikinase inhibitor with Yatiri Bio’s AI-driven ProteoCharts platform, which identified a biomarker-defined AML patient population that may respond to the drug independently of FLT3 status. In May 2026, the company launched a new platform aimed at identifying cancer drug-resistance targets and developing resistance-overcoming oncology therapies.
3. D&D Pharmatech
Location: South Korea
Overall rank: 54
Absolute growth rate (%): 738.41
Compound annual growth rate (%): 103.15
Revenue 2024: $8.39 million
Revenue 2021: $1.19 million
Number of employees: 24
Year founded: 2014
D&D Pharmatech is a South Korea-based biotech focused on GLP-1 and peptide-based therapeutics, with a pipeline spanning obesity, MASH, neurodegenerative disease, fibrotic disease and related indications. The company has built its strategy around long-acting injectable technologies and its Oralink oral peptide platform, which is designed to convert injectable peptide drugs into oral formulations. In April 2026, D&D Pharmatech signed a $1.24 million research contract with Pfizer to develop an oral GLP-1 obesity treatment using Oralink, following Pfizer’s 2025 acquisition of obesity biotech Metsera, D&D’s licensing partner that previously licensed six oral peptide compounds from the company. D&D has also advanced DD01, its GLP-1/glucagon dual agonist for MASH, and will present 48-week Phase II data at EASL Congress 2026. In May 2026, D&D completed a 226.5 billion won ($165 million) convertible bond issuance to support continued clinical and R&D work, including its fibrosis candidate TLY012 and next-generation obesity programs.
4. ABL Bio
Location: South Korea
Overall rank: 82
Absolute growth rate (%): 526.46
Compound annual growth rate (%): 84.34
Revenue 2024: $24.50 million
Revenue 2021: $4.66 million
Number of employees: 104
Year founded: 2016
ABL Bio is a South Korea-based biotech company focused on bispecific antibodies, with a pipeline spanning immuno-oncology, neurodegenerative diseases and antibody-drug conjugates (ADCs). ABL Bio’s business is built around its proprietary Grabody platform, with clinical programs including ABL301/SAR446159, ABL001/tovecimig, ABL111/givastomig and several next-generation bispecific ADC candidates advancing across markets including the US, China, Australia and South Korea. In late 2025, ABL Bio signed a Grabody platform collaboration with Eli Lilly valued at up to $2.60 billion, including a $40 million upfront payment, alongside a $15 million equity investment to support R&D across bispecific antibodies, bispecific ADCs and dual-payload ADCs. On the regulatory side, ABL Bio and NovaBridge received FDA feedback supporting a potential accelerated approval pathway for givastomig in gastric cancer, with a registrational trial targeted as early as Q4 2026, while ABL Bio/NEOK Bio also secured US IND clearance for ABL209, with Phase I trials of ABL206 and ABL209 expected to begin in the first half of 2026.
5. Dx&Vx
Location: South Korea
Overall rank: 123
Absolute growth rate (%): 354.45
Compound annual growth rate (%): 65.64
Revenue 2024: $24.90 million
Revenue 2021: $6.53 million
Number of employees: 121
Year founded: 2001
Dx&Vx is a South Korea-based life sciences company with operations spanning molecular and in vitro diagnostics, healthcare products, companion diagnostics and drug/vaccine development. Its pipeline includes programs in infectious disease, oncology, obesity treatment and eye disease. In July 2025, Dx&Vx signed a co-development and license agreement valued at about $220 million with a US biotech company for its mRNA-based cancer vaccine, marking its first global out-licensing deal. In August 2025, its subsidiary Avixgen signed a separate $360 million licensing-out agreement with a US biotech partner for its ACP peptide-based drug delivery platform, further supporting Dx&Vx’s shift toward platform-driven therapeutics and global partnering.
6. iX Biopharma
Location: Singapore
Overall rank: 178
Absolute growth rate (%): 241.49
Compound annual growth rate (%): 50.59
Revenue 2024: $4.46 million
Revenue 2021: $1.30 million
Number of employees: 59
Year founded: 2004
iX Biopharma is a Singapore-listed specialty pharmaceutical company focused on developing and commercializing therapies built around its proprietary WaferiX sublingual drug delivery platform. iX Biopharma’s portfolio spans pharmaceutical and nutraceutical products, including programs in pain, central nervous system conditions, metabolic disorders and consumer health. Recent company activity has centered on US expansion. In November 2025, iX Biopharma signed a non-binding term sheet with FDA-licensed Orion Specialty Labs to establish a US manufacturing and commercialization base for WaferiX products. And in February 2026, the company received a $41 million US government development contract to support Phase III development and FDA Emergency Use Authorization (EUA)-related work for Wafermine, its lead acute pain program. Wafermine is a patented, sublingual ketamine wafer designed for rapid, non-opioid pain relief, particularly for acute moderate to severe pain. In May 2026, iX announced a restructuring of its consumer business around a new Ligo Pharma vehicle to support its WaferiX-based direct-to-consumer push in the US longevity and lifestyle health market.
7. GNI Group
Location: Japan
Overall rank: 379
Absolute growth rate (%): 86.06
Compound annual growth rate (%): 23.00
Revenue 2024: $155.99 million
Revenue 2021: $115.62 million
Number of employees: 843
Year founded: 2001
GNI Group is a Japan-based life sciences company with operations across Japan, China, the US and Australia, centered on fibrosis-focused pharmaceuticals, drug discovery and medtech. Through its Gyre Therapeutics consolidated subsidiary, GNI Groupthe company markets Etuary (pirfenidone) for idiopathic pulmonary fibrosis (IPF) in China and is advancing F351/hydronidone, a liver fibrosis candidate being developed for chronic hepatitis B-associated liver fibrosis and MASH-related advanced liver fibrosis. In March 2026, Gyre submitted an NDA in China for F351 in CHB-associated liver fibrosis and received priority review from the National Medical Products Administration (NMPA). In May 2026, GNI Group completed an approximately $300 million all-stock acquisition of Cullgen, adding targeted protein degrader and degrader-antibody conjugate programs in inflammatory diseases, cancer and pain.

Comments
Add a comment